Session notes
by Sangjoon Bob Lee
This is a working scratchpad for raw notes taken during microscopy training visits and sessions. Notes here capture practical tips, questions, and observations from hands-on time at the instrument. Over time, useful content gets refined and incorporated into the proper guide sections.
Experiment notes
Gold standard SOP practice: Mar 14, 2026
How to find the ronchigram again?
- Move the joystick around and watch for a small screen current change.
- Then use Diffraction Shift and Focus alignment to find the beam. You will see a faint bit of light.
Remaining questions
- When do you run Stigmator? During real space alignment?
- For C1A1, during the first run, what are the target values? I started with C1 6 nm, A1 27 nm, B2 928 nm. My notes say C1 < 1 nm and A1 < 3 nm. Add target values to the table.
- When do you do manual tuning? With real samples?
- Add a table at the beginning with the target values for aberration corrections.
Open questions
- Ideal Velox play setting? 1024x1024 and 500 ns for gold standard sample.
- When to change condenser stig?
- Camera length and ronchigram size? Proportional: 91 mm to 115 mm, ronchigram size also increases.
- When do you adjust Condenser?
Lessons learned
- Beam condition from FFT: 4-6 rings, each ring with discrete peaks, ~70 pm. Further rings (bigger k) correspond to sharper features resolved in real space.
- Beam condition from Probe corrector: flatness around the “green” aberration surface is the key.
- Overfocus means focal point above the sample; underfocus means below it.
- Defocus change DP? Barely, but real-space probe size is changed (needed for ptycho overlaps).
- Defocus on BF? Expect it to get worse. Ptycho will perform better.
- Focus knob to sharpen? Minimize it. Don’t add degrees of freedom. 20 nm max, use stage piezo and knobs.
- Finding ptycho defocus step size: use ronchigram shadow image to determine feature size variations. Step size of 1 nm is too small.
- Why drift? Inserting the holder itself induces aberrations. After stage movement, find ROI, then wait ~5 min for mechanical stabilization.
Extra notes on aberrations
- Practice getting atomic imaging without Sherpa. Example: Samsung sample, too beam-sensitive for Sherpa.
- Tableau with A5 selected measures up to 5th order aberrations. The following screenshot shows a Tableau measurement result with A5 enabled.

Manual aberration correction without Sherpa (beam-sensitive samples)
- Figure out the full workflow for correcting aberrations without Sherpa.
- Where do you run C1A1 if you can’t expose the sample?
- How do you manually adjust A1 and B2?
- What does “good” look like without Sherpa? Define criteria for the ronchigram, FFT, and probe shape.
- What is the minimum correction quality needed for atomic resolution?
Action steps for future practice sessions
- Try going back to TEM and STEM, confirm aberrations getting worse.
- Try going to LM and then back to regular STEM, confirm resolution getting worse.
- Try correcting the probe without Sherpa, in the case of beam sensitivity.
Cobalt oxide nanoparticles STEM — Mar 3, 2026
It was my first time staying in STEM mode and find samples after STEM probe correction and loading my own sample. The following notes were taken in my attempt to find the feature of interest right after the sample was loaded.
Finding the sample after the sample is loaded
- Go to 5,000× magnification. If there is no beam, it means the beam is blocked on the grid. Move the stage around with the joystick.
- Move the stage until the screen current increases to about 0.150 nA. At this point, the beam has been found. Notice the Kikuchi bands: this is a good clue that you are in a good starting place.
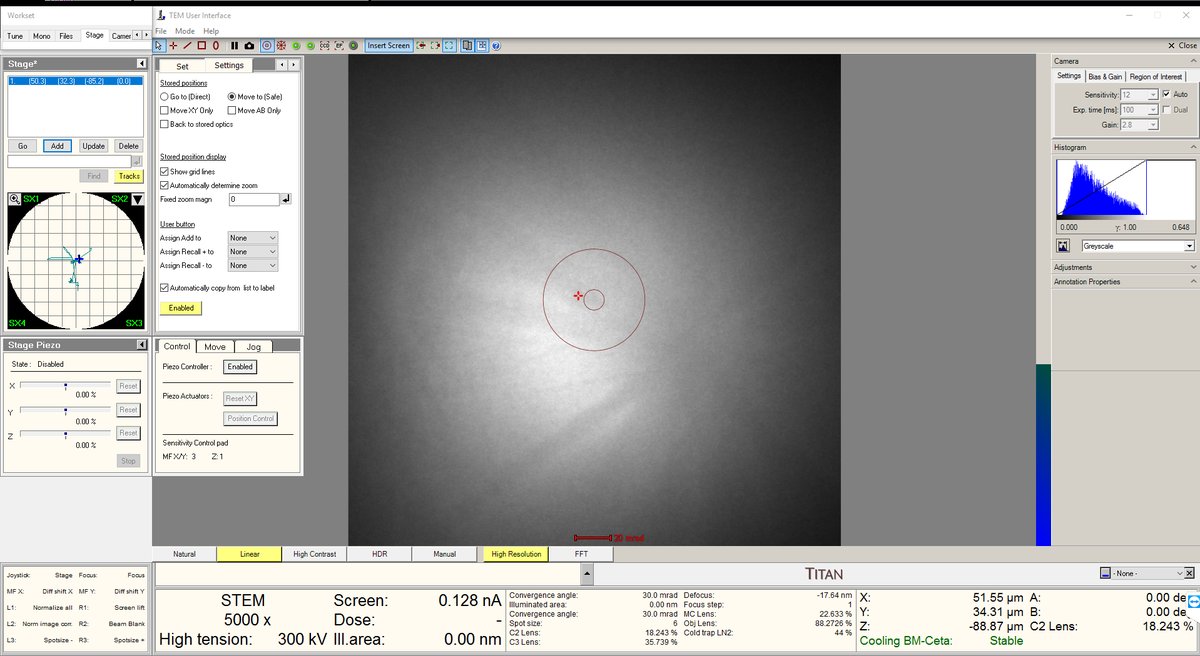
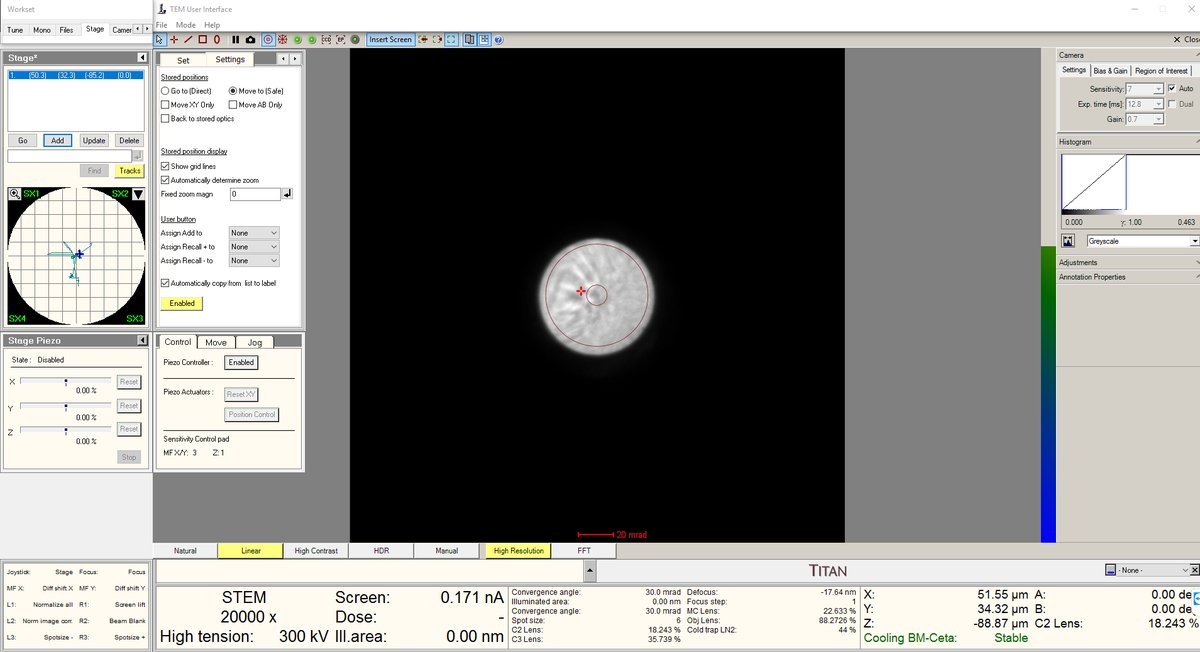
- Increase the magnification to 20,000× or higher. The features will still look blurry since we are not yet at the correct focus.
- Press the Z-axis up and down until the Ronchigram blow-up point appears. Adjust z-axis from 5kx to 20kx to find the blow-up point.
- After the blow-up point, you will see features. In this session, cubes were found after the Ronchigram blow-up point.
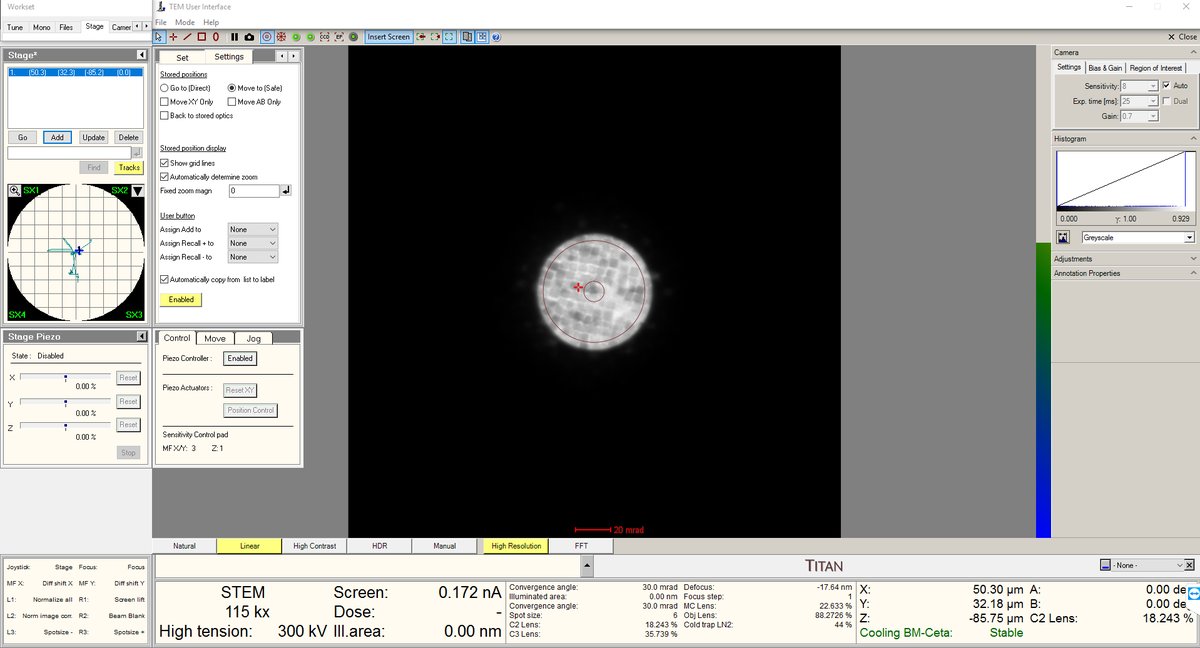
- Stay at the blow-up point. We are now at the eucentric height.
- Turn on the HAADF camera in Velox to observe those features.
Getting sharper features after you’ve found them
- Use the stage piezo to move the sample around to ensure you have the sharpest features.
- Use Sharpa to correct C1A1. You do not need to correct B2A2.
- Take the image as usual.
Notes on aberration correctinos
MAPED experience at NCEM, Mar 2, 2026
I had a chance to join TEAM MAPED session at NCEM with Stephanie Ribet and Henry Bell.
General STEM notes for aberration
- LM mode warning: In TEAM, LM mode isn’t used generally. It turns off the aberration corrector, a set of multipole electromagnetic lenses (hexapoles, octupoles) that correct for spherical aberration. When switched back on, the corrector needs hours to restabilize both thermally (coils heat up, causing alignment drift from thermal expansion) and electromagnetically (currents must settle to precise values).
- aberration knobs: in the probe corrector software, manually fix and it can be faster since software can take a while. For example, Correct A1 (twofold astigmatism) and B2 (axial coma) by hand. (@bobleesj, verifiy this)
- Use the stigmator button on the hand panel, it makes the beam round.
- C2 adjust is used to make the beam concentric, by alinigng C2 aperture.
- Rotation center: don’t care about the edges. Use a magnified image to see whether the features are pulsing out of the page.
Sample loading and vacumm
- After loading a sample, watch PPL. It should go down to low 10⁻³ or 10⁻⁴.
- Octagon must be below 10 after sample loading.
Finding sample ROIs
- In Spectra, you can switch between TEM and STEM modes and it’s stable. On TEAM, this is not the case, so it’s better to use STEM at 5k mag to navigate and find samples.
- Use stage double-click to move around.
Zone axis
- Use alpha and beta on the hand panel to get an approximation, then go to Stage, flap out, and use alpha and beta for fine adjustment.
- Feel free to use camera length to make it easier to see. Ensure the ronchigram is symmetric.
Convergence angle
- Change the convergence angle by changing the aperture.
- C2 for 70 µm aperture gives ~9 mrad max. For higher convergence, use another aperture.
- If you change the C2 aperture, the software may still display the old value (e.g., “20”) because it doesn’t know how to get to the new position. Click “Adjust” to move to the new aperture where it has the intended aperature like 10 micrometer bull’s eye aperture.
- Then move C2 to ~30 (instead of 1,000) to block out other apertures.
Arina at NCEM
- HAADF must be out before you insert Arina. Verify on Digital Micrograph.
- Shutdown order: voltage can be turned off from the software. No need to physically turn it off unlike at SNSF.
Colume valve must be closed even during a quick break
- Always set column valve off for lunch. It does not affect aberrations.
Modify current
- To gain more current, go to Focus and Shift under the
Monotab. This controls the monochromator lens excitation, which determines how tightly the beam is focused at the energy-selecting slit. Lowering focus means a less tight crossover at the slit, so more electrons pass through.
Descan pivot point
- TODO: needs definition and procedure.
TODOs
- Investigate the effect of descan when you integrate or sum across k-space
Reconstruction notes
Ptychography reconstruction basics — Mar 9, 2026
Notes from Arthur on reconstructing data collected from ARINA detector at Stanford.
- Sign convention: in
quantem,C10 > 0means underfocus. The beam focal point is below the sample, hence negative defocus. - Aberrations: SSB is somewhat an “eye test.” One may use the aberrations from SSB or not. There are many degrees of freedom: batch size, the “dose” step size (finer can be better), probe size, center of mass/transpose, and initial aberrations.
- Cropping strategy: in real space, it’s fine to crop, encouraged since faster. In k space, we generally don’t want to crop since we lose the max scattering angle, i.e. we lose fine details in real space.
- Probe: aberrations should be identical across all scan regions in theory. However, for ptycho-tomo, defocus will change with tilt.
- CNN reconstruction: reconstruction weight is different between reconstructions since these are weights being trained.
- Memory: It’s hard to manage memory well in Jupyter notebook but it’s something we can work on.
- Virus samples: there is no zone axis, so we can’t do atomic resolution.
- Descan: the beam is tilted from the source and then tilted back after a short travel perpendicular to the sample. During this second tilt, instability can be introduced and the diffraction pattern isn’t perfectly aligned.
- Mixed probe: mixed probe is good and orthogonality is imposed, so probes should look different from each other.